About GNTI-122
About the Investigational Study Treatment (GNTI-122)
GNTI-122 is an investigational cell therapy being tested in the POLARIS Study. It is manufactured from your own blood cells and is designed to address immune system imbalance associated with Type 1 diabetes.
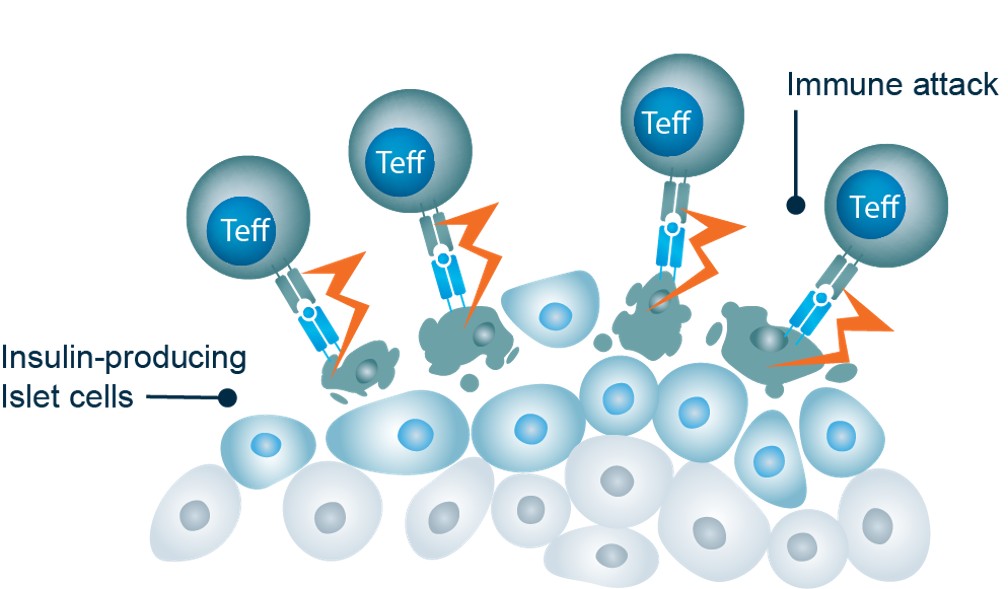
In T1D, effector T cells lose their tolerance for self-antigens expressed on pancreatic beta cells and bind to and destroy these cells.
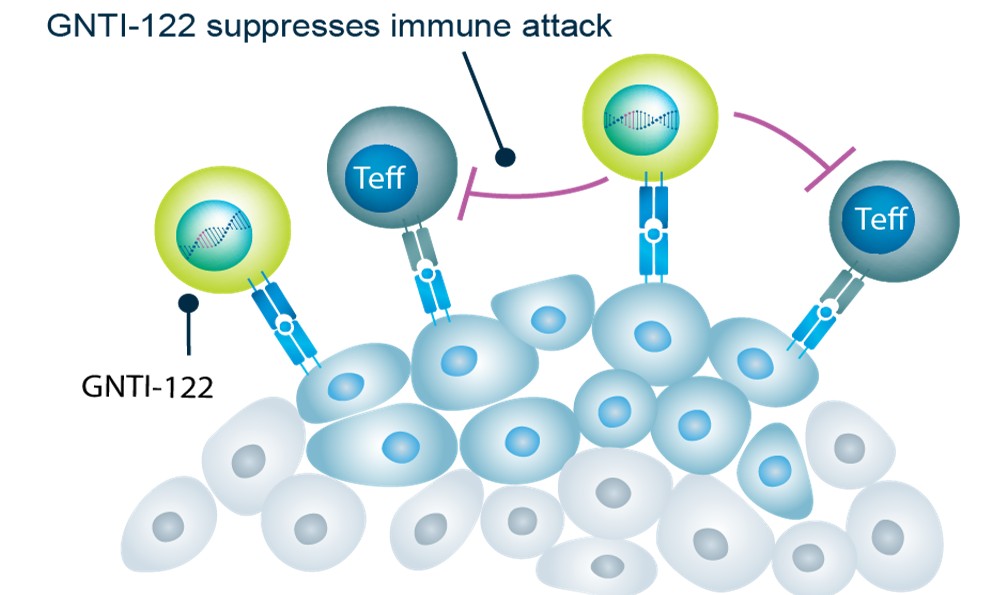
GNTI-122 protects islet cells from damage by homing to the pancreas and draining lymph nodes and suppressing pathogenic effector T cells via the mechanisms of bystander suppression and infectious tolerance.
T regulatory cells, a type of white blood cell important for stopping the destruction of your beta cells in the pancreas. However, T regulatory cells in people with Type 1 Diabetes do not work as well as they should. GNTI-122, an engineered T regulatory cell, is designed to help stop your body from attacking and destroying your beta cells.
GNTI-122 has not been approved by the U.S. Food and Drug Administration (FDA) for general use.